Introduction: CANDOR is a multicenter, phase 3, randomized study of adult patients with relapsed or refractory multiple myeloma (RRMM) previously treated with 1-3 prior lines of therapy (NCT03158688). 466 patients received carfilzomib, dexamethasone, and daratumumab (KdD) or carfilzomib and dexamethasone (Kd) in 2:1 randomization (KdD: 312; Kd: 154). Based on the primary endpoint, KdD demonstrated superior progression-free survival (PFS) vs Kd (hazard ratio [HR], 0.63 [95% CI, 0.46-0.85]; P=0.0014). Deep responses and minimal residual disease (MRD) negativity have been associated with improved PFS for patients with RRMM. Herein, we present an analysis of MRD results from CANDOR.
Methods: Details of the dose and schedule were previously reported (Dimopoulos et al., Lancet 2020). The rate of patients with confirmed CR which were MRD negative (MRD[-]CR) in bone marrow aspirate at 12 months (± 4 weeks) measured by next-generation sequencing (NGS; threshold, 1 tumor cell/10-5 white blood cells) was a prespecified key secondary endpoint. Exploratory analyses included MRD[-]CR at increasing sensitivity (10-4, 10-5, 10-6) and best overall response MRD[-] status at any time point. All reported responses were by Independent Review Committee and were analyzed for the Intent-to-Treat population. MRD[-] status is at <10-5 unless otherwise specified.
Results: The best overall MRD[-]CR rate at any time was 13.8% vs 3.2% in the KdD vs Kd arm (Odds ratio [OR], 4.95; P<0.0001) and the MRD[-] rate regardless of overall response status was 22.8% vs 5.8% (OR, 5.15; P<0.0001). At the 12-month landmark, the MRD[-]CR rate was 12.5% vs 1.3% in the KdD vs Kd arm (OR, 11.3; P<0.0001) and the MRD[-] rate was 17.6% vs 3.9% (OR, 5.76; P<0.0001) with the proportion of patients with MRD[-]VGPR being 4.2% vs 2.6%, respectively. The MRD[-]CR rates at the 12-month landmark for KdD vs Kd were consistent across clinically relevant subgroups (Table).
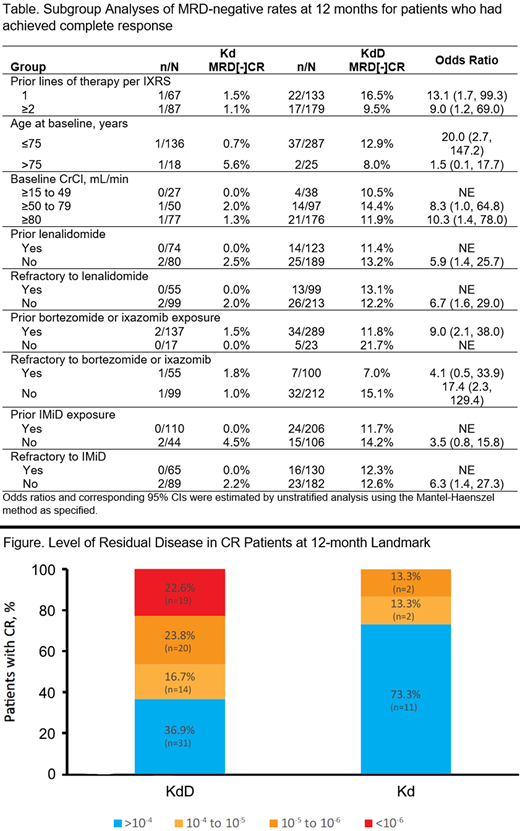
At the 12-month landmark, KdD treatment resulted in a greater proportion of CR rates (26.9% vs 9.7%) and deeper MRD responses than Kd. Among patients in CR, the depth of response as measured by NGS MRD level at the 12-month landmark was deeper for KdD relative to Kd: cutoff of >10-4, 36.9% vs 73.3%; 10-4 to 10-5, 16.7% vs 13.3%; 10-5 to 10-6, 23.8% vs 13.3%; <10-6, 22.6% vs 0% for KdD vs Kd, respectively (Figure). Similar to the results at the 12-month landmark, MRD responses independent of the landmark were deeper among patients in the KdD compared to the Kd arm. With median follow-up of 6 months from the 12-month landmark, no patient with MRD[-]CR response progressed or died.
Additional post hoc analyses were conducted within patients randomized to KdD to explore prognostic characteristics for MRD[-]CR. Importantly, prior lenalidomide exposure did not meaningfully impact the MRD[-]CR rate at the 12-month landmark; 13.2% (25/189), 11.4% (14/123), and 13.1% (13/99) for naïve, exposed, and refractory subgroups, respectively. For prior bortezomib, the MRD[-]CR rates were 24% (6/25), 11.5% (33/287), and 6.8% (6/88) for naïve, exposed, and refractory subgroups, respectively. The rates of MRD[-]CR at the 12- month landmark within the KdD arm were consistent across subgroups: patients refractory to the last prior therapy (yes vs no, 10.9% vs 14.3%), number of prior regimens (1-2 vs 3 prior regimens; 13.2% vs 10.1%), prior transplant (yes vs no, 11.8% vs 13.7%), duration of first remission (≤2 vs >2 years, 12.3% vs 13% and ≤1 vs >1 year, 10.7% vs 13.4%), baseline creatinine clearance (≥15 to <50, ≥50 to <80, and ≥80 mL/min, 10.5%, 14.4%, and 11.9%, respectively), age (≤75 vs >75 years, 12.9% vs 8.0%), or dose intensity (< vs ≥ median) for carfilzomib or daratumumab (10.5% vs 14.9% and 9.8% vs 15.6%, respectively). Data on cytogenetics will be included at the time of presentation.
Conclusion: At the primary analysis, patients treated with KdD achieved significantly higher MRD[-]CR rates vs Kd at the 12-month landmark. Among patients with an MRD[-]CR, the depth of MRD was deeper with KdD vs Kd. With a median of 6 months follow-up, no patient with an MRD[-]CR has progressed; duration of response will be updated at time of presentation. Within the KdD arm, lenalidomide exposure or refractoriness did not diminish the MRD[-]CR rate. These findings support the efficacy of the KdD regimen as an effective treatment for RRMM, including patients who have become lenalidomide refractory.
Landgren:Adaptive: Consultancy, Honoraria; Amgen: Consultancy, Honoraria, Research Funding; Pfizer: Consultancy, Honoraria; Juno: Consultancy, Honoraria; Cellectis: Consultancy, Honoraria; BMS: Consultancy, Honoraria; Binding Site: Consultancy, Honoraria; Karyopharma: Research Funding; Merck: Other; Pfizer: Consultancy, Honoraria; Seattle Genetics: Research Funding; Juno: Consultancy, Honoraria; Glenmark: Consultancy, Honoraria, Research Funding; Cellectis: Consultancy, Honoraria; BMS: Consultancy, Honoraria; Takeda: Other: Independent Data Monitoring Committees for clinical trials, Research Funding; Binding Site: Consultancy, Honoraria; Karyopharma: Research Funding; Janssen: Consultancy, Honoraria, Other: Independent Data Monitoring Committees for clinical trials, Research Funding; Seattle Genetics: Research Funding; Celgene: Consultancy, Honoraria, Research Funding; Glenmark: Consultancy, Honoraria, Research Funding; Takeda: Other: Independent Data Monitoring Committees for clinical trials, Research Funding; Janssen: Consultancy, Honoraria, Other: Independent Data Monitoring Committees for clinical trials, Research Funding; Celgene: Consultancy, Honoraria, Research Funding; Merck: Other. Weisel:Roche: Consultancy, Honoraria; Amgen: Consultancy, Honoraria, Research Funding; Celgene: Consultancy, Honoraria, Research Funding; Janssen: Consultancy, Honoraria, Research Funding; Sanofi: Consultancy, Honoraria, Research Funding; Bristol-Myers Squibb: Consultancy, Honoraria; Adaptive: Consultancy, Honoraria; GlaxoSmithKline: Honoraria; Karyopharm: Consultancy, Honoraria; Takeda: Consultancy, Honoraria; Abbvie: Consultancy, Honoraria. Rosinol Dachs:Janssen: Honoraria; Celgene: Honoraria; Amgen: Honoraria; Takeda: Honoraria; Sanofi: Honoraria. Moreau:Sanofi: Consultancy, Honoraria; Amgen: Consultancy, Honoraria; Celgene/Bristol-Myers Squibb: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; Novartis: Honoraria; Takeda: Honoraria; Abbvie: Consultancy, Honoraria. Hajek:PharmaMar: Consultancy, Honoraria; Takeda: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Abbvie: Consultancy, Honoraria; Oncopeptides: Consultancy; Novartis: Consultancy, Research Funding; Janssen: Consultancy, Honoraria, Research Funding; Amgen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; BMS: Consultancy, Honoraria, Research Funding; Celgene: Consultancy, Honoraria, Research Funding. Mollee:Janssen: Membership on an entity's Board of Directors or advisory committees, Research Funding; BMS/Celgene: Membership on an entity's Board of Directors or advisory committees; Amgen: Membership on an entity's Board of Directors or advisory committees; Takeda: Membership on an entity's Board of Directors or advisory committees; Pfizer: Membership on an entity's Board of Directors or advisory committees; Caelum: Membership on an entity's Board of Directors or advisory committees. Kim:Alexion Pharmaceuticals Inc.: Honoraria, Research Funding. Zhang:Amgen: Current Employment. Go:Amgen: Current Employment. Morris:Amgen: Current Employment. Usmani:Celgene: Other; Takeda: Consultancy, Honoraria, Other: Speaking Fees, Research Funding; Janssen: Consultancy, Honoraria, Other: Speaking Fees, Research Funding; SkylineDX: Consultancy, Research Funding; Seattle Genetics: Consultancy, Research Funding; Merck: Consultancy, Research Funding; Incyte: Research Funding; Pharmacyclics: Research Funding; Array Biopharma: Research Funding; GSK: Consultancy, Research Funding; Sanofi: Consultancy, Honoraria, Research Funding; Abbvie: Consultancy; BMS, Celgene: Consultancy, Honoraria, Other: Speaking Fees, Research Funding; Amgen: Consultancy, Honoraria, Other: Speaking Fees, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal